Sep 20, 2021
Paingone
Topical Lidocaine Treatment Recalled by Manufacturer
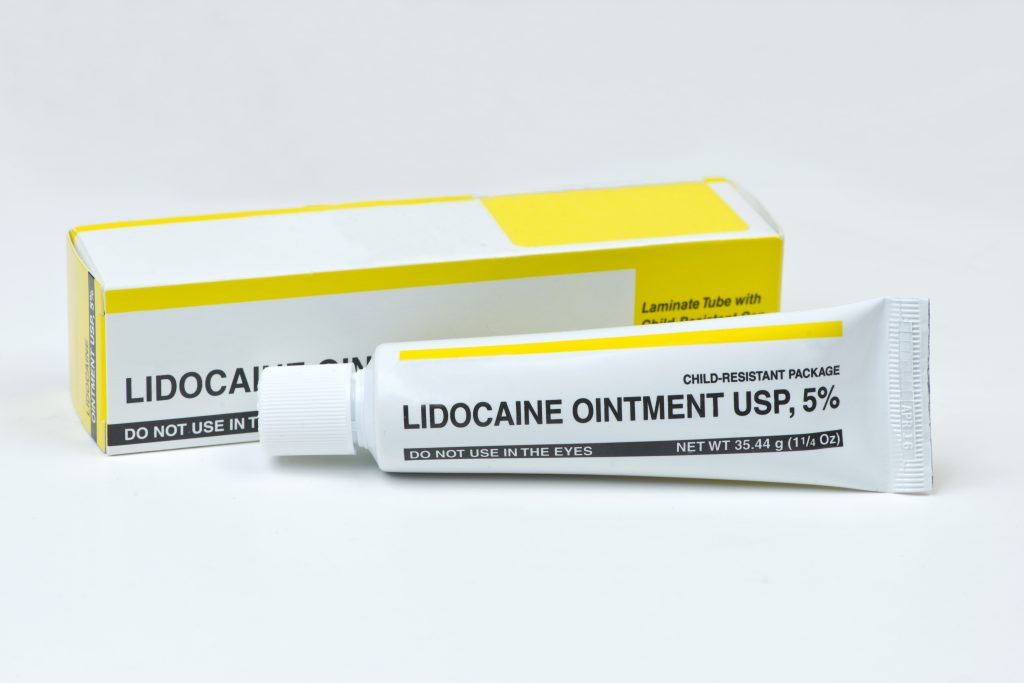
Topical lidocaine lotions and sprays can provide temporary pain relief, but a recent recall by Teligent Pharma highlights the potential dangers of lidocaine solutions. The company has voluntarily recalled their Lidocaine HCI Topical Solution (4%), which is sold in screw cap glass bottles.
The lidocaine product was recalled for super potency, which could lead to anesthetic toxicity or death if not treated promptly. So far, there have been no reports of death or severe injury from the recalled products. The lot under recall is Lidocaine Topical Solution 4% (40mg/mL); NDC# 52565-009-50; Lot# 14218; Exp 9/2022. It was originally distributed through both retailers and wholesale outlets in Canada and the United States.
If you have used this product or had any adverse reactions to it, contact your physician. You can also call Teligent at 856-697-1441 or email medical@teligent.com if you have any questions.
FDA Grants Orphan Drug Status to Treatment for Complex Regional Pain Syndrome (CRPS)
Soin Therapeutics has been granted approval by the FDA to create an investigational form of low-dose naltrexone, which is currently used to treat alcohol and prescription drug abuse. While it works at current dosages to relieve cravings and euphoria in those who abuse alcohol and medications, lower doses may help relieve pain.
According to a press release from Soin, naltrexone “has also been shown in anecdotal reports to alleviate symptoms stemming from CRPS at very low doses (approximately one tenth the dose for OUD) of currently approved indications. When the dose of the drug is substantially lowered, the pharmacological effects change favorably to treat CRPS.”
Soin Therapeutics is formulating a novel, low-dose version of naltrexone in anticipation of submitting an Investigational New Drug Application to the FDA. They hope to gain approval for a large-scale, national clinical trial in the future.
Liquid Filled Aspirin Capsules Now Available Over the Counter
A liquid-filled aspirin capsule is now available in the United States under the brand name Vazalore. PLx Pharma manufactures the capsules. Vazalore was approved by the FDA in March of this year. The OTC medication is recommended for individuals with rheumatic and vascular diseases undergoing aspirin therapy. The liquid form targets the release of active ingredients within the digestive tract in order to improve absorption.
Vazalore absorbs much faster (up to four times as fast) than enteric-coated aspirin caplets, with an absorption rate nearly five times as high. Patients had significantly less erosion of the stomach and intestinal tract during clinical trials when using the liquid-filled capsules instead of traditional, rapid-release aspirin tablets.
Over 50% of U.S. Physicians Recommend Complementary Healthcare
The Journal of Alternative and Complementary Healthcare published a report indicating that office-based physicians and healthcare providers in the United States are increasingly recommending various forms of complementary healthcare, including alternative treatments for acute and chronic pain.
The study noted that female physicians are more likely to recommend alternative treatments than male doctors. Over 63% of women doctors recommend complementary approaches over 12 months, while 49.3% of men did so. The most commonly recommended complementary treatments for pain include massage therapy, chiropractic adjustment, yoga, and acupuncture.
The study results indicate an increasing interest in complementary and alternative care approaches by physicians across a number of specialties. Most doctors in the study indicated their patients had positive results when adding complementary healthcare to their more traditional, medication-based treatments.
A novel, Protein-Based Approach to Treating Neuropathic Pain Shows Promise
Researchers at Monash University, Australia, have announced the discovery of a unique treatment for neuropathic pain, including diabetic neuropathy, that could be an alternative to opioids. The non-opioid analgesics seem to be safe and effective against even severe neuropathy. The study results were recently published in the journal Nature.
The study was carried out by the University’s Monash Institute of Pharmaceutical Sciences (MIPS) and the Monash Biomedicine Discovery Institute (BDI). Research revealed that targeting a particular receptor protein (adenosine A1) can be successful in developing an effective pain reliever. In the past, targeting the pain receptor protein was deemed too difficult, but recent advances rely on a specific type of molecule, a positive allosteric modulator (PAM), that is highly targeted and binds to a particular region of the adenosine A1 protein.
Associate Professor Wendy Imlach, a co-corresponding author of the study and head of the Pain Mechanisms lab at BDI, explained, “This study has helped us to better understand mechanisms underpinning allosteric drug actions. One of the exciting things we found is that not only were the PAMs able to decrease neuropathic pain with minimal unwanted effects, but they actually increase their level of effectiveness as the pain signals in the spinal cord get stronger – thus highlighting the potential for allosteric medicines that are uniquely sensitive to disease context”.
This Week’s Pain Fact: Recently, a report issued by the U.S. Centers for Disease Control and Prevention National Center for Health Statistics revealed that in the three months prior to a research study, approximately 60% of Americans reported some form of pain requiring treatment of some kind. In order of frequency, the top three types of pain were lower back pain, leg pain, and pain in the upper extremities.
Is Paingone helping your patients and your practice?
Tell us your success story.
A robust retail strategy is essential to growing your practice.
You only have so many hours a week available for appointments. The fastest way to increase your bottom line is to incorporate profitable products that generate repeat business and build your reputation as a medical professional. That’s where Paingone comes in.
Contact us below to learn more about our products, wholesale pricing and how Paingone can benefit your practice.

